Laboratory of Electrostatic Methods of Bioencapsulation
Head of the Laboratory
Assoc. Prof. Dorota Lewińska, DSc. PhD,
From 2022, after joining the Team of prof. Andrzej Chwojnowski, the research topics conducted at LEMB have been expanded and now cover three basic research topics. three basic research topics
Pierwszy z nich ścisłe związany jest z wykorzystaniem impulsowego pola elektrostatycznego do wytwarzania polimerowych nośników substancji biologicznie aktywnych oraz żywych komórek. Obejmują one następujące rodzaje nośników:
Polymer nonwovens.
Currently conducted work includes, among others, research on the influence of pulse electrospinning process parameters on the structure of the produced nonwovens. Using multiparametric factor analysis type 23, the following were investigated:
- influence of selected electrospinning process parameters (voltage, polymer solution flow rate and polymer solution viscosity) on fibre thickness and the ability of the system to produce bead-on-string (Fig. 1a ) and homogeneous (Fig. 1b) nonwovens.

Rys. 1. Zdjęcia SEM włóknin polimerowych o różnej morfologii: a) typu bead-on string, b) jednorodnych.
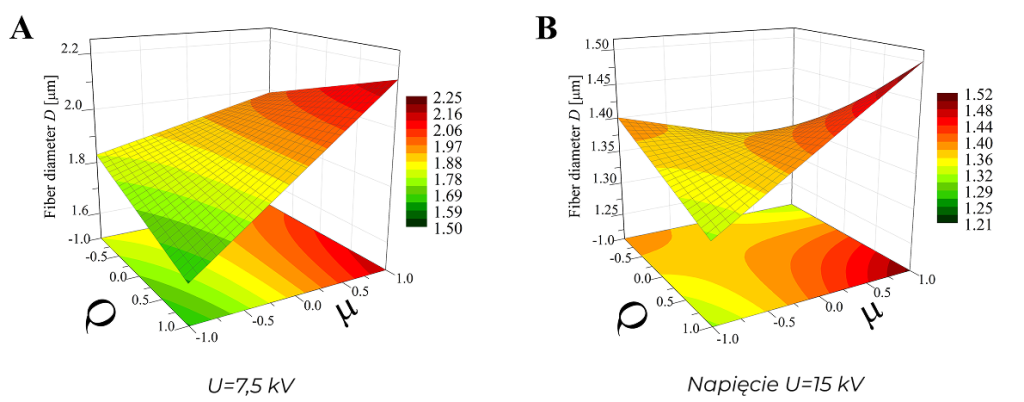
Fig. 2. Example result of a factor analysis: surface plots showing the effect of the polymer solution flow rate Q (in the range 0.6-1.2 ml/h) and the viscosity of this solution µ (in the range 0.043-0.107 Pa*s) on the fibre thickness D for homogeneous nonwovens produced at different voltage values U. Korycka et al. Beilstein J. Nanotechnol. 2018.
- Wpływ wielkości parametrów elektrycznych procesu elektroprzędzenia impulsowego (U, f i τ) na średnicę i morfologię mono- i bi-modalnych włóknin polimerowych.


Rys. 3. Zdjęcie SEM włókniny: a) monomodalnej o jednkowej grubości nici i b) bi-modalnej, zbudowanej z nici o różnej grubości. Bartkowiak et al. Polymers 2024.
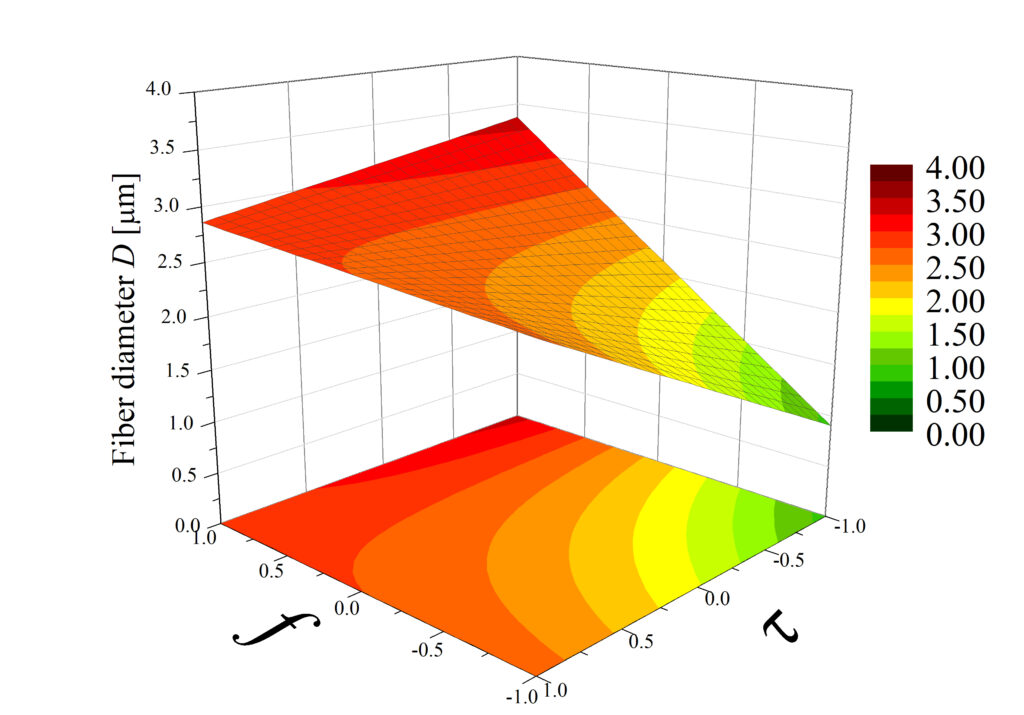
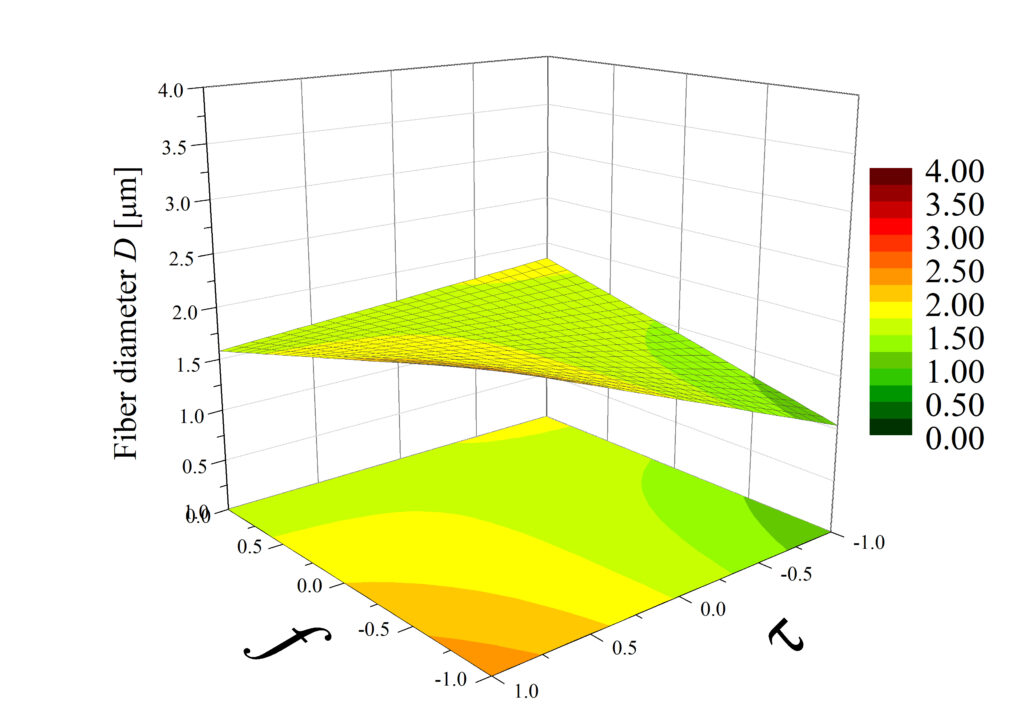
Rys. 4. Przykładowy wynik analizy czynnikowej: wykresy powierzchniowe przedstawiające wpływ częstotliwości impulsów elektrycznych f (w zakresie 30-100 Hz) i czasu trwania tych impulsów (w zakresie 1-9 ms) na grubość włókien D, wytworzonych przy różnych wartościach napięcia U. Bartkowiak et al. Polymers 2024.
Sferyczne mikrokulki polimerowe
Sferyczne mikrokulki polimerowe z polimerów syntetycznych i naturalnych przeznaczone do immobilizacji leków i żywych komórek.

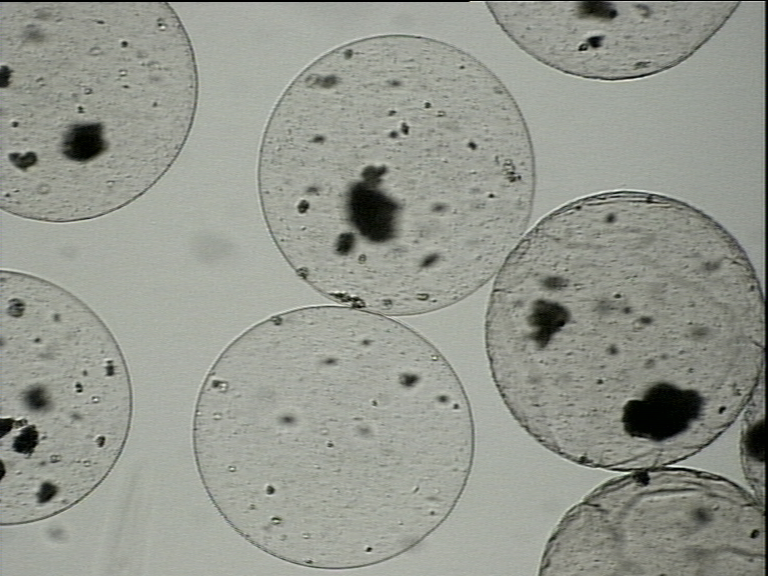
Rys. 5. Sferyczne mikromatryce polimerowe wytworzone z: a) polimeru syntetycznego Mirek et al. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2022; b) polisacharydu zawierające immobilizowane komórki ludzkich przytarczyc.
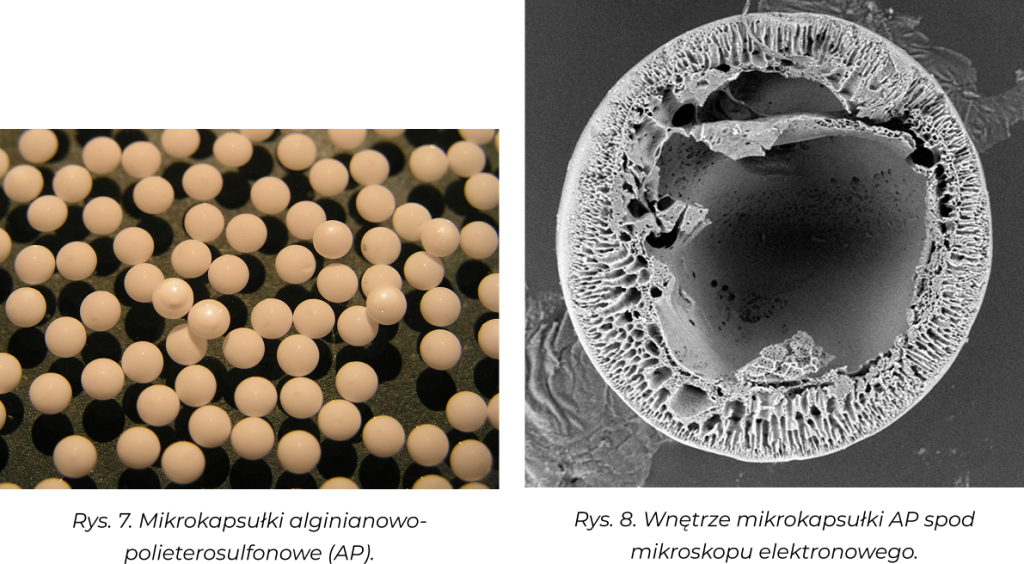
Alginate and alginate/polyethersulfone microcapsules, designed for encapsulation of living cells, manufactured in a single-stage process using coaxial heads:
- double-nozzle - RP PATENT No. 208384
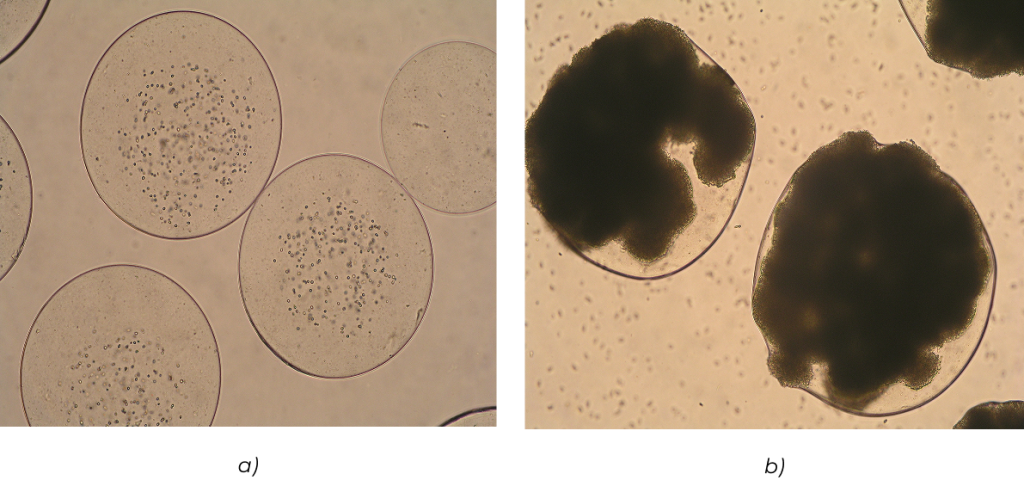
Rys. 6. Komórki drożdży piekarskich enkapsulowane wewnątrz sferycznych matryc alginianowych wytworzonych za pomocą głowicy dwudyszowej: a) bezpośrednio po wytworzeniu, b) po 48 godzinach hodowli. Lewińska et al. Biocybernetics and Biomedical Engineering, 2008, 28(2), 69-84
- • three-nozzle - (hydrogel core/polymer semi-permeable membrane made of synthetic polymer PATENT RP No. 208383
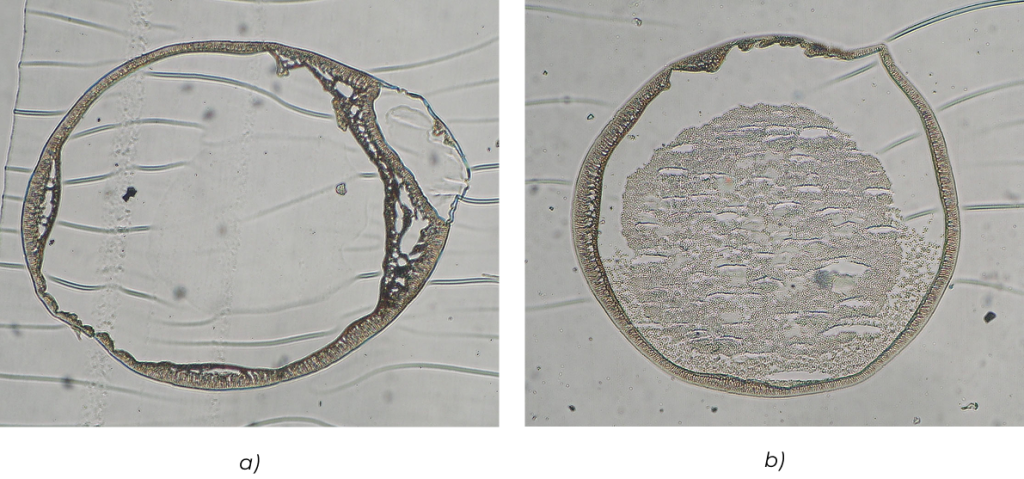
Rys. 9. Wnętrze mikrokapsułek AP zawierających enkapsulowane drożdże piekarskie: (a) przed i (b) po 24 godz. hodowli.
The Laboratory also conducts work on developing methods for testing the properties of manufactured polymer carriers Lewińska et al. J. Membrane Sci, Grzeczkowicz et al. Desalination and Water Treatment 2018, Kupikowska-Stobba et al. Biocybernetics and Biomedical Engineering 2021, and recently also develops inks for 3D bioprinting Mirek et al. Biomaterials Advances, 2023.
Keywords: electrospinning, electrospraying, pulsed electrostatic field, microcapsules, microbeads, cell microencapsulation.
Selected publications:
- Korycka P., Mirek A., Kramek-Romanowska K., Grzeczkowicz M., Lewińska D. „Effect of electrospinning process variables on the average size of polymer fibers and bead-on-string structures established with a factorial design.” Beilstein J. Nanotechnol. 2018, 9, 2466-2478
- Kramek-Romanowska K., Grzeczkowicz M., Korycka P., Lewińska D. “A Factorial Design for Assessment of the Effect of Selected Process Variables on the Impulse Electrostatic Droplet Formation.” In: Korbicz J., Maniewski R., Patan K., Kowal M. (eds) Current Trends in Biomedical Engineering and Bioimages Analysis. PCBEE 2019. Advances in Intelligent Systems and Computing, vol 1033, pp 327-336, (2020) Springer, Cham, DOI https://doi.org/10.1007/978-3-030-29885-2_30
- Bartkowiak A., Grzeczkowicz M., Lewińska D. “The Effects of Pulsed Electrospinning Process Variables on the Size of Polymer Fibers Established with a 23 Factorial Design” Polymers 2024, 16, 2352. DOI https://doi.org/10.3390/polym16162352
- Mirek A., Grzeczkowicz M., Lamboux C., Syreina Sayegh S., Bechelany M., Lewińska D. “Formation of disaggregated polymer microspheres by a novel method combining pulsed voltage electrospray and wet phase inversion techniques.” Colloids and Surfaces A: Physicochemical and Engineering Aspects, Vol. 648, (2022), 129246, DOI: https://doi.org/10.1016/j.colsurfa.2022.129246
- Rosiński S., Lewińska D., Migaj M., Woźniewicz B., Weryński A. “Electrostatic microencapsulation of parathyroid cells as a tool for the investigation of cells activity after transplantation”; Landbauforschung Voelkenrode, Special Issue, 241, 47-50, 2002
- Lewińska D., Bukowski J., Kożuchowski M., Kinasiewicz A., Weryński A. “Electrostatic microencapsulation of living cells”. Biocyb and Biomed Eng, vol. 28(2), 69-84, 2008
- Jasińska U., Skąpska S., Owczarek L., Dekowska A., Lewińska D. „Immobilization of Bifidobacterium infantis cells in selected hydrogels as a method of increasing their survival in fermented milkless beverages” Journal of Food Quality, vol. 2018, doi.org/10.1155/2018/9267038
- Cendrowski P., Kramek-Romanowska K., Lewińska D., Grzeczkowicz M., Korycka P., Krzysztoforski J. “CFD modeling of droplet generation process for medical applications using the electrostatic impulse method” Chemical and Process Engineering 2022, 43 (3), 331–355., DOI: 10.24425/cpe.2022.142278
- Lewińska D., Chwojnowski A., Wojciechowski C., Kupikowska-Stobba B., Grzeczkowicz M., Weryński A. „Electrostatic droplet generator with 3-coaxial-nozzle head for microencapsulation of living cells in hydrogel covered by synthetic polymer membranes” Sep Sci and Technol, vol. 47(3), 463-469, 2012
- Lewińska D., Grzeczkowicz M., Kupikowska-Stobba B. “Influence of electric parameters on the alginate-polyethersulfone microcapsule structure” Desalination and Water Treatment vol. 64, 400-408, 2017 DOI: 10.5004/dwt.2017.11407. 64 (2017)
- Przytulska M., Kulikowski J. L., Lewińska D., Grzeczkowicz M., Kupikowska-Stobba B. „Computer-Aided Image Analysis for Microcapsules’ Quality Assessment.” Biocybernetics and Biomedical Engineering, vol. 35(4):342-350, 2015, DOI:10.1016/j.bbe.2015.05.005
- Mirek A., Korycka P., Grzeczkowicz M. Lewińska D.,(2019) “Polymer fibers electrospun using pulsed voltage” Materials and Design 183 (2019) 108106, DOI: 10.1016/j.matdes.2019.108106
- Mirek A., Grzeczkowicz M., Belaid H., Bartkowiak A., Barranger F., Abid M., Wasyłeczko M., Pogorielov M., Bechelany M., Lewińska D., “Electrospun UV-cross-linked polyvinylpyrrolidone fibers modified with polycaprolactone/polyethersulfone microspheres for drug delivery” Biomaterials Advances 147 (2023) 213330, https://doi.org/10.1016/j.bioadv.2023.213330
- Kupikowska-Stobba B., Grzeczkowicz M., Lewińska D., “A one-step in vitro continuous flow assessment of protein release from core-shell polymer, microcapsules designed for therapeutic protein delivery.” Biocybernetics and Biomedical Engineering, vol. 41 (2021) 1347– 1364, 2021, DOI https://doi.org/10.1016/j.bbe.2021.05.003
- Kupikowska-Stobba B., Lewińska D., “Polymer microcapsules and microbeads as cell carriers for in vivo biomedical applications”, Biomaterials Science, 2020, DOI: 10.1039/c9bm01337g
- Lewińska D., Rosiński S., Hunkeler D., Poncelet D., Weryński A. “Mass transfer coefficient in characterization of gel beads and microcapsules” J. Membrane Sci, vol. 209(2): 533-540, 2002
- Hunkeler D., Wandrey Ch., Rosiński S., Lewińska D., Weryński A. Characterization of microcapsules (in: Fundamentals of Cell Immobilisation Biotechnology, eds. Nedovič V, Willaert R), Kluwer Academic Publishers, Dordrecht, Boston, London, 8A, 389-410, 2004
- Grzeczkowicz M., Lewińska D. “A method for investigating transport properties of partly biodegradable spherical membranes using vitamin B12 as marker” Desalination and Water Treatment, 128:170-178, 2018 , doi: 10.5004/dwt.2018.22631
- Kupikowska-Stobba B., Lewińska D., Grzeczkowicz M. „Chemical method for retrieval of cells encapsulated in alginate-polyethersulfone microcapsules”, Artificial Cells, Nanomedicine, and Biotechnology vol. 42(3): 151-160, 2014 DOI:10.3109/21691401.2013.800083
- Lewińska D., Rosiński S., Weryński A. “Influence of process conditions during impulsed electrostatic droplet formation on size distribution of hydrogel beads” Artif Cell, Blood Substitutes and Biotechnol, vol. 32, 1, 41-53, 2002
- Prusse U., Bilancetti L., Bucko M., Bugarski B., Bukowski B., Gemainer P., Lewińska D., Massart B., Nastruzzi C., Nedovic V., Poncelet D., Siebenhaar S., Tobler L., Tosi A., Vikartovska A., Vorlop K-D. “Comparison of different technologies for alginate bead production”. Chem Papers, vol. 62(4), 364-374, 2008
- Kupikowska B., Lewińska D., Dudziński K., Jankowska-Śliwińska J., Grzeczkowicz M., Wojciechowski C. “The influence of changes in composition of membrane-forming solution on the structure of alginate-polyethersulfone microcapsules”. Biocyb and Biomed Eng, vol. 29(3), 61-69, 2009
- Mirek A., Belaid H., Barranger F., Grzeczkowicz M., Bouden Y., Cavaillès V., Lewińska D, Bechelany M., “Development of a new 3D bioprinted antibiotic delivery system based on a cross-linked gelatin–alginate hydrogel.” J. Mater. Chem. B, 2022, Advance Article, DOI https://doi.org/10.1039/D2TB01268E
- Mirek A., Belaid H., Bartkowiak A., Barranger F., Salmeron F., Kajdan M, Grzeczkowicz M., Cavaill`es V., Lewinska D., Bechelany M. „Gelatin methacrylate hydrogel with drug-loaded polymer microspheres as a new bioink for 3D bioprinting.”, Biomaterials Advances 150 (2023) 213436, DOI: https://doi.org/10.1016/j.bioadv.2023.213436
Patents:
- Korycka P., Mirek A., Lewińska D., Grzeczkowicz M., Chwojnowski A: “Method of manufacturing biodegradable polymer membrane.” PATENT RP No. 237648, 2021
- Lewińska D., Kupikowska-Stobba B., Grzeczkowicz M., Chwojnowski A., Łukowska E.: „Sposób oznaczania stężenia komórek.” PATENT RP Nr 223717, 2012
- Lewińska D., Chwojnowski A., Jankowska-Śliwińska J., Weryński A.: „Mikrokapsułki zawierające substancje biologicznie aktywne, zwłaszcza żywe komórki i/lub mikroorganizmy, ewentualnie genetycznie modyfikowane, albo naturalne lub syntetyczne do zastosowań biomedycznych, sposób ich wytwarzania oraz urządzenie do stosowania tego sposobu”, . PATENT RP Nr 208383, 2007
- Lewińska D., Bukowski J., Rosiński S,. Kożuchowski M., Weryński A.: ”Sposób jednoetapowego wytwarzania mikrokapsułek, zwłaszcza zawierających żywe komórki, ich zbiory lub substancje biologicznie aktywne, oraz urządzenie do ich wytwarzania”, PATENT RP Nr 208384, 2004
International cooperation::
- W latach 2020-2024 współpraca z Universite de Montpellier we Francji w ramach umowy pt.:”International Join Doctorate Convention” zawartej pomiedzy w/w instytutem a IBIB PAN w dniu 30.04.2020 r. dotycząca współprowadzenia przewodu doktorskiego mgr inż. Adama Mirka
- W latach 2005-2007 w ramach dwustronnego projektu Polonium współpraca z grupą prof. Cecile Legallais z University of Technology, Compiegne, CNRS, UMR 6600, Compiegne Francja w temacie: „Modification of alginate beads with hepatocytes for bioartificial liver.”
- 1999 - 2004 direct scientific cooperation within the frame of the European Project COST 840 Action “Bioencapsulation. Innovations and Technologies” with research groups from:
- Thüne Insitute of Agricultural Technology, Braunschweig, Germany;
- Institute of Chemistry Slovak Academy of Sciences, Bratislava, Slovakia;
- Deptartment of Chemical Engineering Faculty of Technology and Metallurgy, Belgrade, Serbia and Montenegro;
- ENITIAA, UMR CNRS GEPEA, Nantes, Francja;
- Laboratory of Polyelectrolytes and Biomacromolecules, Swiss Federal Institute of Technology, Lausanne, Switzerland;
- Institute of Pharmaceutical Technology and Biopharmaceutics, University of Vienna, Austria;
School of Chemical Engineering, University of Birmingham, Edgbaston, Birmingham, Great Britain; - Laboratory of Chemical and Biological Engineering, Swiss Federal Institute of Technology, Lausanne, Switzerland;;
- Department of Pharmacy and Pharmaceutical Technology, University of Basque Country, Vitoria, Spain.
Grants:
- “The elaboration of the method of structural homogenous membrane preparation in alginate-polyethersulfone microcapsule applying the computer-aided image analysis methods.” 29-month grant No 4151/B/T02/2009/36 funded by Polish State Committee for Scientific Research 2009 - 2011.
- “Developing and studying a modified electrostatic microcapsule generator. Performing transport studies of microcapsules produced within the COST 840 project” Special Grant SPUB-M No. 56/E-83/ SPUB/COST/P-04/DZ584/2002-2004 funded by Polish State Committee for Scientific Research realized between 2002-2004
“A new therapeutic strategy in transplantology. Allotransplantation of human parathyroid cells in vitro in therapy of hypoparathyroidism without immunosuppression” Research project PB 538/PO5/98/15 funded by Polish State Committee for Scientific Research realized between 1998-2001
- “COST 840 Action – Selection of conditions in the process of microcapsule formation using an electrostatic field and coating microcapsules in membrane, and development of a method for studying their transport properties in vitro.” Special Grant SPUB-M No. 56/E-83/ SPUB/COST/P-04/DZ 62/99 funded by Polish State Committee for Scientific Research realized between 1998-2001
The development of novel polymeric cell scaffolds for articular cartilage regeneration.
The second research topic conducted in the EMB Laboratory, led by Prof Andrzej Chwojnowski, concerns the development of novel polymeric cell scaffolds for articular cartilage regeneration.
Why are we doing it?
Every year the number of patients suffering from cartilage damage is growing. These injuries significantly reduce the patients' quality of life. Currently, there is no effective method for cartilage regeneration. One of the most promising approaches is the use of scaffolds. They provide support for cartilage cells (chondrocytes) or stem cells, promoting their development and regeneration.
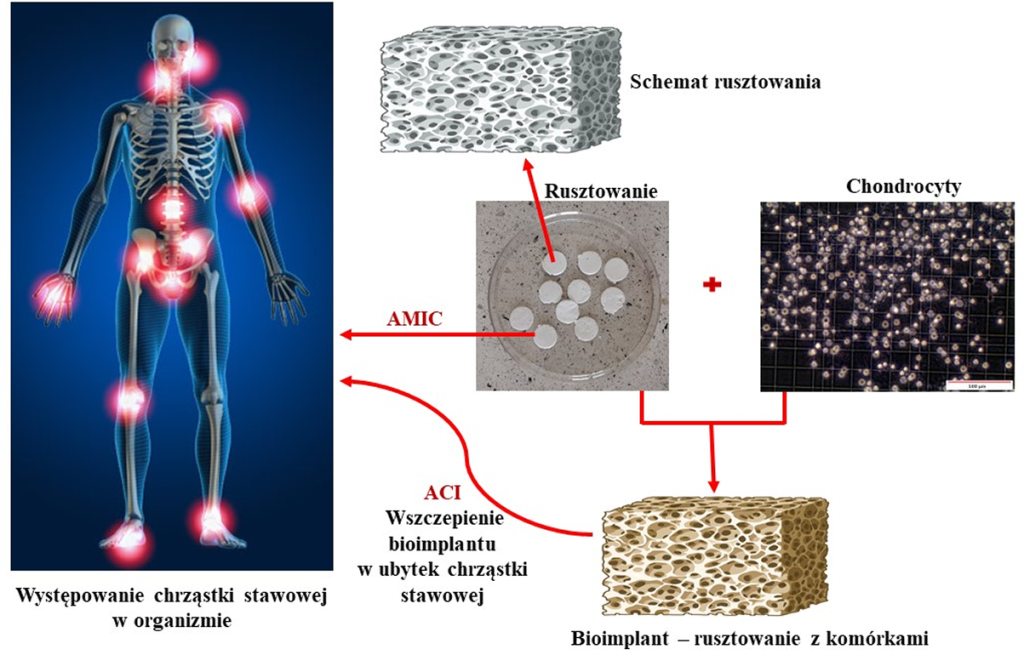
Rys. 10. Występowanie chrząstki stawowej w ogranizmie oraz schemat metod terapeutycznych z wykorzystaniem rusztowań komórkowych do regeneracji chrząstki stawowej.
How do we work?
In our laboratory, we specialize in designing and obtaining cellular scaffolds that can be used in the most promising therapeutic approaches, such as ACI (Autologous Chondrocyte Implantation) and AMIC (Autologous Matrix-Induced Chondrogenesis). We are developing scaffolds with controlled porosity, bioactivity and biodegradability to best support articular cartilage regeneration [1].
Each developed scaffold undergoes testing including:
- Structure analysis - imaging with a scanning electron microscope (SEM) and advanced MeMoExplorer software, which enables accurate analysis and evaluation of pore geometry and distribution, and results with which data can be obtained regarding the reproducibility of scaffold structures [2]. In our research, we focus on obtaining scaffolds that allow the culture of stem cells and chondrocytes, tailored to their specific requirements. Adequate pore size is essential to obtain scaffolds that can be used for effective therapy with available ACI and AMIC methods. To ensure optimal conditions for cell growth and differentiation, we design scaffolds with the appropriate pore size. For stem cells, scaffolds with pores above 300 µm, while chondrocytes require smaller pores, below 300 µm.
The resulting scaffolds, despite the use of different polymers, are characterized by reproducibility and varying pore sizes [3-6].
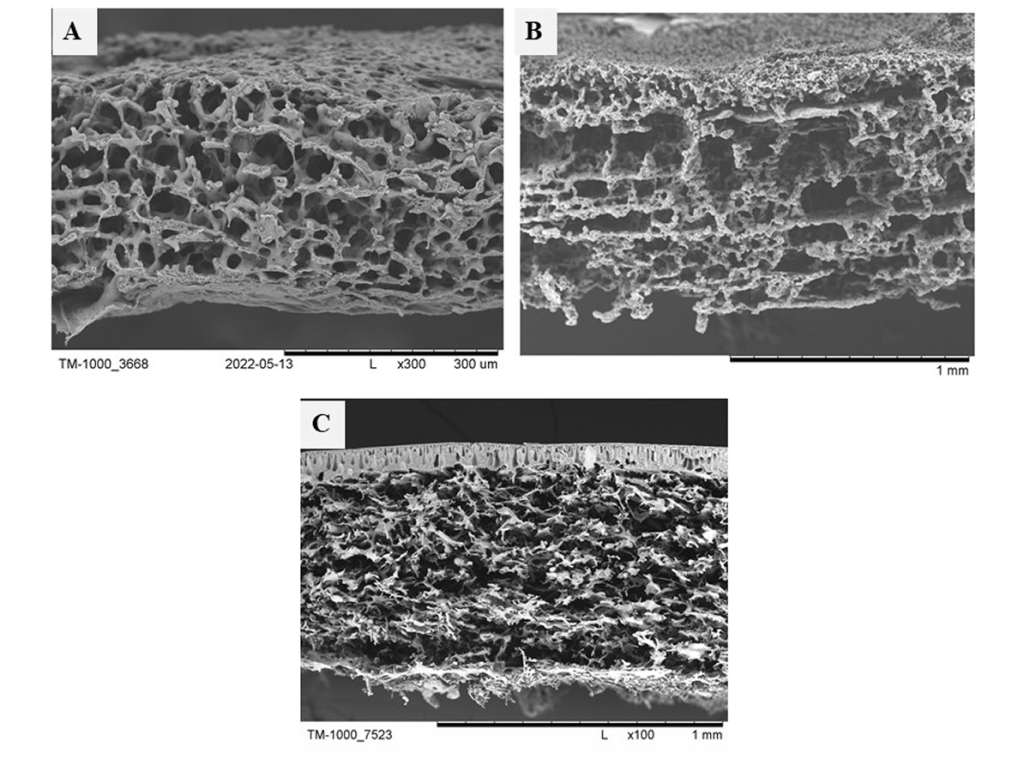
Rys. 11. Obrazy SEM rusztowań zaprojektowanych i otrzymanych w Laboratorium Membran Półprzepuszczalnych. Rusztowanie zostały otrzymane z: A – mieszaniny polimerów PES i PUR; B – kopolimeru polilaktyd-ko-kaprolakton; C – z polieterosulfonu [3-6].
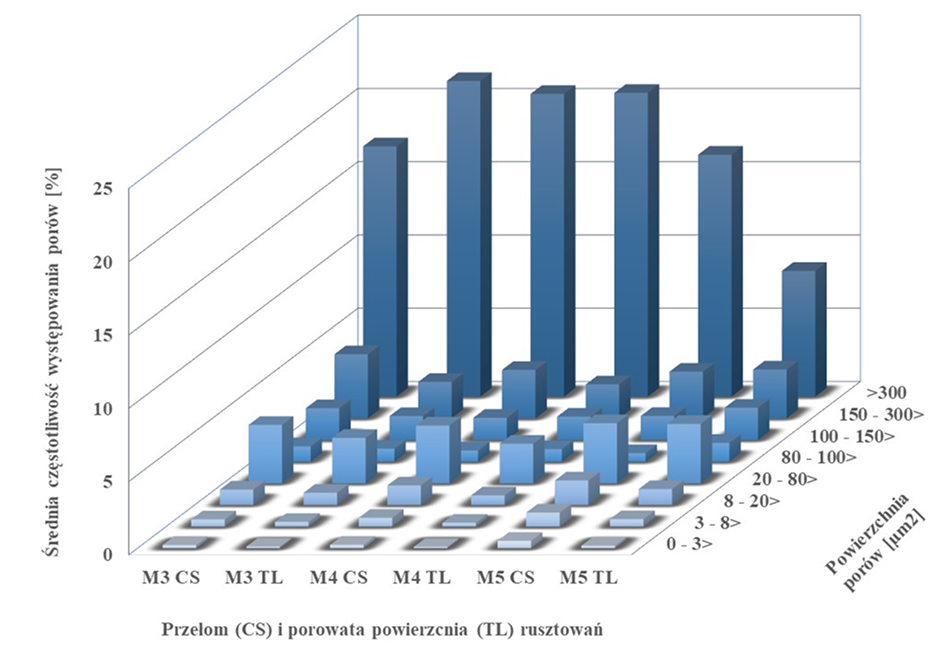
Fig. 12. The diagram shows the average frequency of pores in eight pore size classes in scaffold cross-section
(CS) and scaffold top layer (TL).
The diagram was made using MeMoExplorer software.
- Mechanical properties - crucial for joint biomechanics, including strength and flexibility [3];
- Porosity and wettability - essential for cell colonization and nutrient transport [3];
- Bio-degradation - using physiological fluids, such as In Simulated Body Fluid (SBF) [3], [4] .
In Vitro and In Vivo studies
The most promising scaffolds were further validated:s
- In vitro - tests using isolated human chondrocytes to assess the scaffold's ability to promote cell proliferation and differentiation [6].
- In vivo - selected scaffolds were tested on a rabbit model in collaboration with orthopedic surgeons from Gruca University Hospital for Orthopaedics and Traumatology, Department of Traumatology and Orthopaedics in Otwock [7-9].
If you would like to learn more about our research and scaffolding or establish cooperation, please feel free to contact us!
Selected publications:
- M. Wasyłeczko, W. Sikorska, A. Chwojnowski, Review of synthetic and hybrid scaffolds in cartilage tissue engineering, Membranes. 10 (2020) 1–28. https://doi.org/10.3390/membranes10110348.
- M. Przytulska, J.L. Kulikowski, M. Wasyłeczko, A. Chwojnowski, D. Piętka, The evaluation of 3D morphological structure of porous membranes based on a computer-aided analysis of their 2D images, Desalination and Water Treatment. 128 (2018). https://doi.org/10.5004/dwt.2018.22569.
- M. Wasyłeczko, E. Remiszewska, W. Sikorska, J. Dulnik, A. Chwojnowskl, Scaffolds for Cartilage Tissue Engineering from a Blend of Polyethersulfone and Polyurethane Polymers, Molecules (Basel, Switzerland). (2023) 1–25. https://doi.org/10.3390/molecules28073195.
- M. Wasyłeczko, W. Sikorska, M. Przytulska, J. Dulnik, A. Chwojnowski, Polyester membranes as 3D scaffolds for cell culture, Desalination and Water Treatment. 214 (2021) 181–193. https://doi.org/10.5004/dwt.2021.26658.
- K. Dudziński, A. Chwojnowski, M. Gutowska, M. Płończak, J. Czubak, E. Łukowska, C. Wojciechowski, Three dimensional polyethersulphone scaffold for chondrocytes cultivation – The future supportive material for articular cartilage regeneration, Biocybernetics and Biomedical Engineering. 30 (2010) 65–76.
- M. Wasyłeczko, Z.J. Krysiak, E. Łukowska, M. Gruba, W. Sikorska, A. Kruk, J. Dulnik, J. Czubak, A. Chwojnowski, Three-dimensional scaffolds for bioengineering of cartilage tissue, Biocybernetics and Biomedical Engineering. 42 (2022) 494–511. https://doi.org/10.1016/j.bbe.2022.03.004.
- M. Baranowski, M. Wasyłeczko, A. Kosowska, A. Plichta, S. Kowalczyk, A. Chwojnowski, W. Bielecki, J. Czubak, Regeneration of Articular Cartilage Using Membranes of Polyester Scaffolds in a Rabbit Model, Pharmaceutics. 14 (2022). https://doi.org/10.3390/pharmaceutics14051016.
- M. Płończak, M. Wasyłeczko, T. Jakutowicz, A. Chwojnowski, Intraarticular Implantation of Autologous Chondrocytes Placed on Collagen or Polyethersulfone Scaffolds : An Experimental Study in Rabbits, (2023) 4–7.
- T. Jakutowicz, M. Wasyłeczko, M. Płończak, C. Wojciechowski, A. Chwojnowski, J. Czubak, Comparative Study of Autogenic and Allogenic Chondrocyte Transplants on Polyethersulfone Scaffolds for Cartilage Regeneration, 2024. https://doi.org/10.3390/
Patents:
- A. Chwojnowski, E, Łukowska, C. Wojciechowski, M. Wasyłeczko, W. Sikorska, Z. Krysiak, „Sposób wyodrębniania białka z hodowli komórkowych prowadzonych na rusztowaniach komórkowych”. PATENT PL 238140, 2021
- A. Chwojnowski, E. Łukowska, C. Wojciechowski, M. Wasyłeczko, W. Sikorska, Z. Krysiak, „Sposób wykrywania pozostałości celulozy w półprzepuszczalnych membranach szeroko porowatych”. PATENT PL 235794, 2020
- E. Łukowska, A. Szakiel, M. Markowski, M. Wasyłeczko, A. Chwojnowski, S. Martyniuk. “Multilayer polyester membrane and the method of its manufacture”. PATENT PL No 239461
3. Semi-permeable, hollow-fiber polymeric membranes with various properties
Within the framework of the third research direction, semi-permeable, hollow-fiber polymeric membranes with various properties tuned to specific needs in biomedical (e.g., cell culture) and biotechnological (e.g., “anti-fouling” membranes) applications are being developed, fabricated and studied.
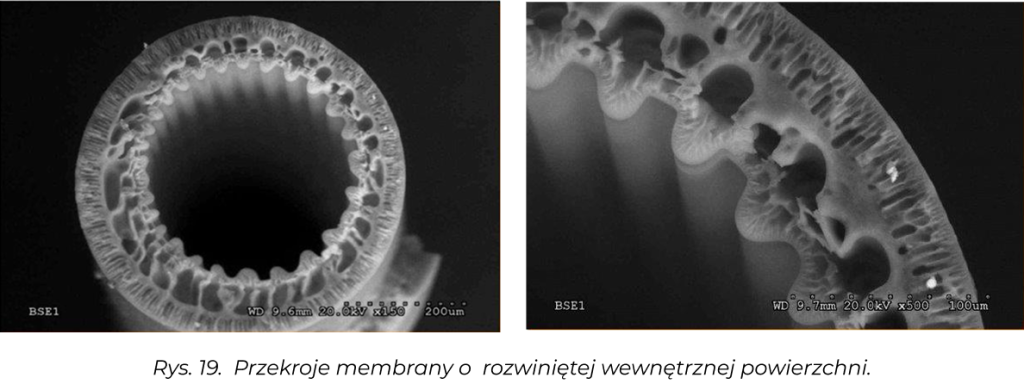
The membranes are manufactured by spinning on an installation developed by the Team for obtaining hollow-fiber membranes using the wet phase inversion method by extruding a polymer solution through a through a capillary-forming spinneret. We produce polysulfone (PSf), polyethersulfone (PES), cellulose acetate (CA), polyurethane (PU) and mixed membranes. By changing the parameters of membrane spinning, we have the ability to obtain membranes with the desired transport-separation parameters [1].


A method has also been developed for producing duo-layer membranes, e.g. PSf/PU, PSf/OC, obtained by means of a special spinneret, in which one polymer forms the outer layer and the other the inner layer and these polymers do not mix with each other.
Degradable multi-component zbudowane są z polimeru bazowego stanowiącego trwałą konstrukcje membrany i polimeru degradowalnego, usuwanego ze struktury membran w czasie ich pracy. Mimo zachodzącego foulingu (zatykania porów membrany przez separowane bądź oczyszczane substancje) usuwany stopniowo polimer zwiększa porowatość membrany, co wydłuża czas jej pracy. Polimerem bazowym jest najczęściej PSf lub PES, polimerami degradowalnymi są: OC [3], polilaktyd, polikaprolakton, poliglikolid, kopolimer laktyd-glikolit-kaprolakton) [4] oraz PU [5-8].
Transport and separation properties membran takie jak przepuszczalność hydrauliczna (współczynnik ultrafiltracji UFC), retencje (wyznaczanie punktu odcięcia) oraz porowatość struktury membran określana za pomocą analizy zdjęć SEM [9-11], przeprowadzonej z użyciem opracowanego w naszym Instytucie programu komputerowego MeMoExplorerTM [12].
We collaborate with other laboratories of our Institute by developing and fabricating membranes and membrane mini-modules for use in bioreactors [13-15] and cell cultures.
An important activity of our laboratory is didactics. Studenci Politechniki Warszawskiej realizują u nas prace inżynierskie, magisterskie i doktorskie. Prowadzone są ćwiczenia i praktyki studenckie.
Selected publications:
- C. Wojciechowski, A. Chwojnowski, K. Dudzinski, E. Lukowska. Evaluation of separation characteristic of polysulfone membranes modified by polymer solvents etching. Desalination and Water Treatment, vol 35, 263-270, 2011.
- A. Chwojnowski, C. Wojciechowski, K. Dudziński, E. Łukowska. Polysulphone and polyethersulphone hollow fiber membranes with developed inner surface as material for bio-medical applications. Biocybernetics and Biomedical Engineering. Vol. 29, No. 3, 47-59, 2009
- C. Wojciechowski, A. Chwojnowski, L. Granicka, E. Łukowska. Polysulfone/cellulose acetate blend semi degradable capillary membranes preparation and characterization. Desalination and Water Treatment. vol 64, 365-371, 2017
- C. Wojciechowski, M. Mazurek-Budzyńska, A. Palinska, A. Chwojnowski, L.Granicka, W. Sikorska, G. Rokicki. Preparation and characterization of partially degradable hollow fiber membranes based on polysulfone/poly(L-lactide-co-glycolide-co-ε-caprolactone) blends. Desalination and Water Treatment. vol 202, 38-47, 2020
- C. Wojciechowski, A. Chwojnowski, L. Granicka, E. Łukowska, M. Grzeczkowicz. Polysulfone/polyurethane blend degradable hollow fiber membranes preparation and transport-separation properties evaluation. Desalination and Water Treatment. vol 57, 22191-22199, 2016
- W. Sikorska, C. Wojciechowski, M. Przytulska, G. Rokicki, M. Wasyłeczko, J. Kulikowski, A. Chwojnowski. Polysulfone–polyurethane (PSf-PUR) blend partly degradable hollow fiber membranes: preparation, characterization, and computer image analysis. Desalination and Water Treatment. vol 128, 383-391, October 2018
- W. Sikorska, M. Wasyłeczko, M. Przytulska, C. Wojciechowski, G. Rokicki, A. Chwojnowski. Chemical Degradation of PSF‐PUR Blend Hollow Fiber Membranes—Assessment of Changes in Properties and Morphology after Hydrolysis. Membranes.vol. 11, 51, 12 January 2021
- W. Sikorska, M. Milner-Krawczyk , M. Wasyłeczko, C. Wojciechowski, A. Chwojnowski. Biodegradation Process of PSF-PUR Blend Hollow Fiber Membranes Using Escherichia coli Bacteria—Evaluation of Changes in Properties and Porosity. Polymers. 13, 1311, 16 April 2021
- A. Chwojnowski, C. Wojciechowski, D. Lewińska, E. Łukowska, J. Nowak, B. Kupikowska- Stobba, M. Grzeczkowicz. Studies on the structure of semi-permeable membranes by means of SEM. Problems and potential sources of errors. Biocybernetics and Biomedical Engineering, vol 32, numb.1, 51-64, 2012.
- A. Chwojnowski, M. Przytulska, D. Wierzbicka, J. Kulikowski, C. Wojciechowski. Membranes’ porosity evaluation by komputer-aided analysis of SEM images – A preliminary study. Biocybernetics and Biomedical Engineering. vol 32, numb.4, 65-75, 2012
- M. Przytulska, A. Kruk, J. Kulikowski, C. Wojciechowski, A. Gadomska-Gajadhur, A. Chwojnowski. Comparative Assessment of Polyvinylpirrolidone. Type of membranes. Based on porosity analysis. Desalination and Water Treatment. vol 75, 18–25, May 2017
- Sikorska W., Przytulska M., Wasyłeczko M., Wojciechowski C., Kulikowski J.L., Chwojnowski A. Influence of hydrolysis, solvent and PVP addition on the porosity of PSF/PUR blend partly degradable hollow fiber membranes evaluated using the MeMoExplorer software. Desalination and Water Treatment. 214, 114–119, February 2021
- A. Ciechanowska, P. Ładyżyński, G. Hoser, S. Sabalińska, J. Kawiak, P. Foltyński, C. Wojciechowski, A. Chwojnowski. Human endothelial cells hollow fiber membrane bioreaktor as a model of the blood vessel for in vitro studies. Journal of Artificial Organs. vol. 19, Issue 3, pp 270–277, Sept. 2016
- 1K. Pluta, M. Jakubowska; M. Wisniewska; A. Wencel; C. Wojciechowski; M. Gora; K. Dudek; A. Chwojnowski; B. Burzynska; D. Pijanowska. Hollow Fiber Bioreactor with Genetically Modified Hepatic Cells as a Model of Biologically Active Function Block of the Bioartificial Liver. Biocybernetics and Biomedical Engineering. Vol. 44(1), 9-19, 2023(4)
- P. Ładyżynski, A. Ciechanowska, S. Sabalinska, P. Foltynski, A. Wencel, C. Wojciechowski, K. Pluta, A. Chwojnowski. Development of an Artificial Blood Vessel Model in a Capillary Bioreactor and Assessment of the Effect of Glucose Levels on HUVECs Under Physiological Shear Stress, Biocybernetics and Biomedical Engineering. 44, 543–559, 2024